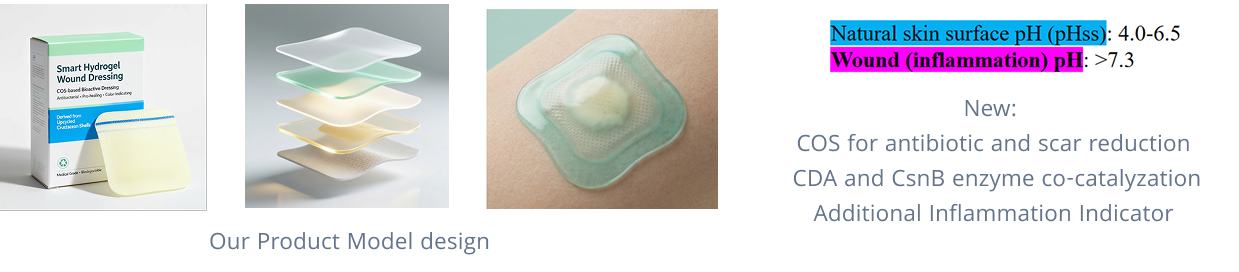
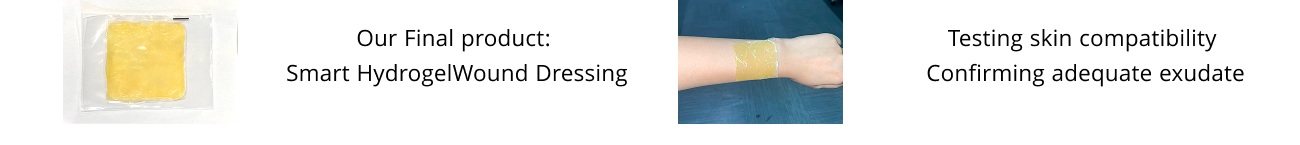
Our project originated from a simple observation: coastal cities generate large amounts of discarded shrimp and crab shells. Further exploration revealed that these shells are rich in chitin, a valuable biopolymer with proven benefits in infection control, wound healing, and scar reduction.
While advanced wound care products face challenges in balancing these functions, the seafood industry continues to produce massive shell waste. Our work bridges these two gaps by converting crustacean shell waste into a sustainable, high-value medical material for advanced wound care.